Telehealth is Here to Stay: New 50 State Survey of Commercial Insurance Laws Reveals Progress
December 2, 2019

Two years after Foley’s 2017 Telemedicine and Digital Health Survey uncovered a demand for telemedicine and digital health services, as well as coverage and reimbursement to match the growing demand, Foley explores how each state has responded in its 2019 50-State Telehealth Survey plus District of Columbia (DC) of Commercial Payer Statutes.
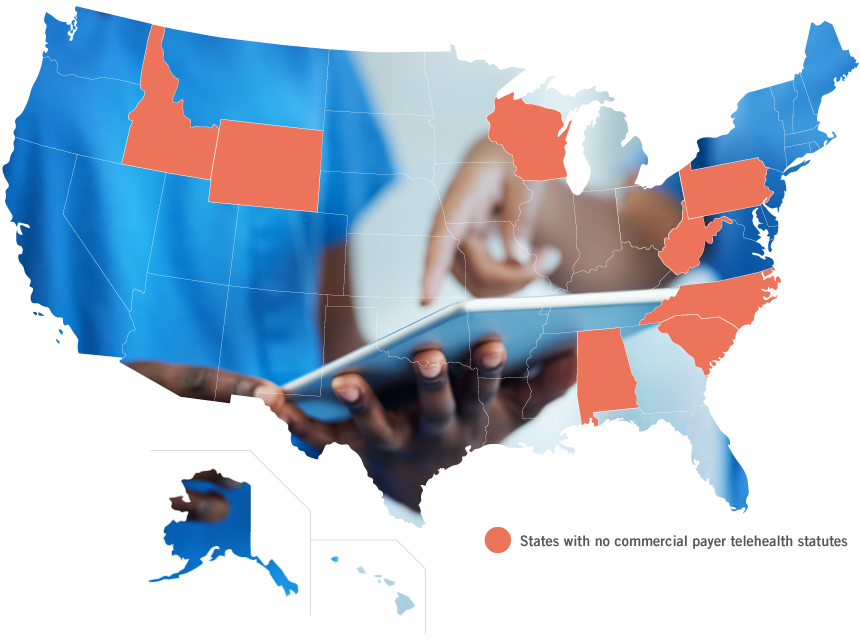
The survey found that as of October 2019, 42 states maintain some sort of telehealth commercial payer law, however several of these state laws do not actually require coverage or payment parity.
Foley’s report breaks down variances in telehealth commercial statues state-by-state, including:
- States that have commercial payer statutes in place
- States that have coverage parity or limitations
- States that have payment parity
- States that impose originating site limits
- States that have patient cost shifting protection
- State laws that address telehealth narrow or exclusive networking
- States that require coverage of reimbursement of remote patient monitoring
- States that require coverage of store & forward/asynchronous telemedicine
To read the full survey, download the 2019 report.
Author(s)
Related Insights
December 29, 2025
Tariff & International Trade Resource
Mexican January 2026 Tariff Tsunami: Maquilas Aren’t Immune
On January 1, 2026, Mexico will increase its general import tariff rate (known as the most favored nation (MFN) rate). The increase will be in the range of five to fifty percent, impacting 1,463 eight-digit tariff lines encompassing thousands of products originating in countries with which Mexico does not have a free trade agreement (FTA or the measure).
December 24, 2025
Health Care Law Today
Gender-Affirming Care: Multi‑State Lawsuit Challenges HHS Declaration
As previously discussed in Foley’s healthcarelawtoday, on December 18, 2025, the U.S. Department of Health & Human Services (HHS) held a press conference focused on what it defined as “sex rejection procedures” (SRPs), also known as gender-affirming care (GAC) for minors, and outlined next steps.
December 23, 2025
Energy Current
FERC Opens New Paths for Co-Located Loads in PJM: What Data Center and Power Generation Developers Need to Know
Key Takeaways FERC has ordered PJM to overhaul its tariff framework for co-located generation and large loads, finding existing rules…

